21 CFR Part 11

In the pharmaceutical and biotechnology industries, ensuring data integrity, security, and traceability is paramount. To establish a set of criteria for electronic records and signatures, the US Food and Drug Administration (FDA) introduced 21 CFR Part 11. This regulation outlines the requirements for using electronic records and signatures in place of traditional paper-based systems in FDA-regulated industries.
Understanding and implementing 21 CFR Part 11 compliance can be challenging, but it is essential for organizations that wish to maintain regulatory approval and avoid potential legal consequences. This article provides an overview of 21 CFR Part 11 compliance and offers guidance for navigating the complex requirements.
Overview of FDA 21 CFR Part 11
21 CFR Part 11, or Title 21 of the Code of Federal Regulations, Part 11, establishes criteria for electronic records and electronic signatures to be considered trustworthy, reliable, and equivalent to paper records and handwritten signatures. The regulation applies to any organization that submits information to the FDA in electronic form or that wishes to maintain electronic records in lieu of paper records.
- Subpart A – General Provisions
- Subpart B – Electronic Records
- Subpart C – Electronic Signatures
Key Components of FDA 21 CFR Part 11 Compliance
- Validation: Organizations must demonstrate that their electronic systems are capable of producing accurate, consistent, and reliable results. This process typically involves validating software applications, infrastructure, and hardware systems.
- Audit Trails: The regulation requires that electronic records include a secure, computer-generated, time-stamped audit trail. This must document the date and time of operator entries and actions that create, modify, or delete electronic records.
- Data Security and Integrity: Organizations must implement technical and procedural controls to ensure the integrity and confidentiality of electronic records. This includes data encryption, access controls, and backup and recovery processes.
- Electronic Signatures: To be considered valid, electronic signatures must be unique to the individual, linked to the corresponding electronic record, and require two distinct identification components (such as a user ID and password).
- Training: Employees who use electronic systems must receive appropriate training to ensure they understand the system’s functionality and their responsibilities under 21 CFR Part 11.
Implementing FDA 21 CFR Part 11 Compliance
- Perform a Gap Analysis: Identify any shortcomings in the organization’s current processes and systems, and develop a plan to address these gaps.
- Establish a Cross-Functional Team: Assemble a team of key stakeholders from various departments, including IT, Quality Assurance, and Regulatory Affairs, to oversee the compliance project.
- Develop and Implement Policies and Procedures: Create a comprehensive set of policies and procedures that outline how the organization will achieve and maintain compliance with 21 CFR Part 11.
- Conduct Employee Training: Ensure all employees who use electronic systems are trained on the new policies, procedures, and system functionality.
- Perform Regular Audits: Regularly assess and audit electronic systems to ensure ongoing compliance and address any potential issues.
Achieve Compliance with testRigor Automated Testing
- Use plain English commands to outline test steps: Describe each action and requirement in simple terms to ensure that the process is easily understandable by all stakeholders.
- Compile a comprehensive PDF report: Consolidate your compliance documentation, including screenshots and actions taken at each step, into a single PDF file for easy reference and submission to regulatory authorities.
Login click "Patient Registration click "Add New Patient" generate unique name, then enter into "First Name" and save as "newPatientName" generate unique email, then enter into "Email" and save as "newPatientEmail" click "Submit" check that page contains "Added successfully." enter saved value "newPatientName" into "search" enter enter check that page contains saved value "newPatientEmail"
Below are screenshot examples from a PDF report, compliant with 21 CFR Part 11 (we’re using a simple test project for this example):
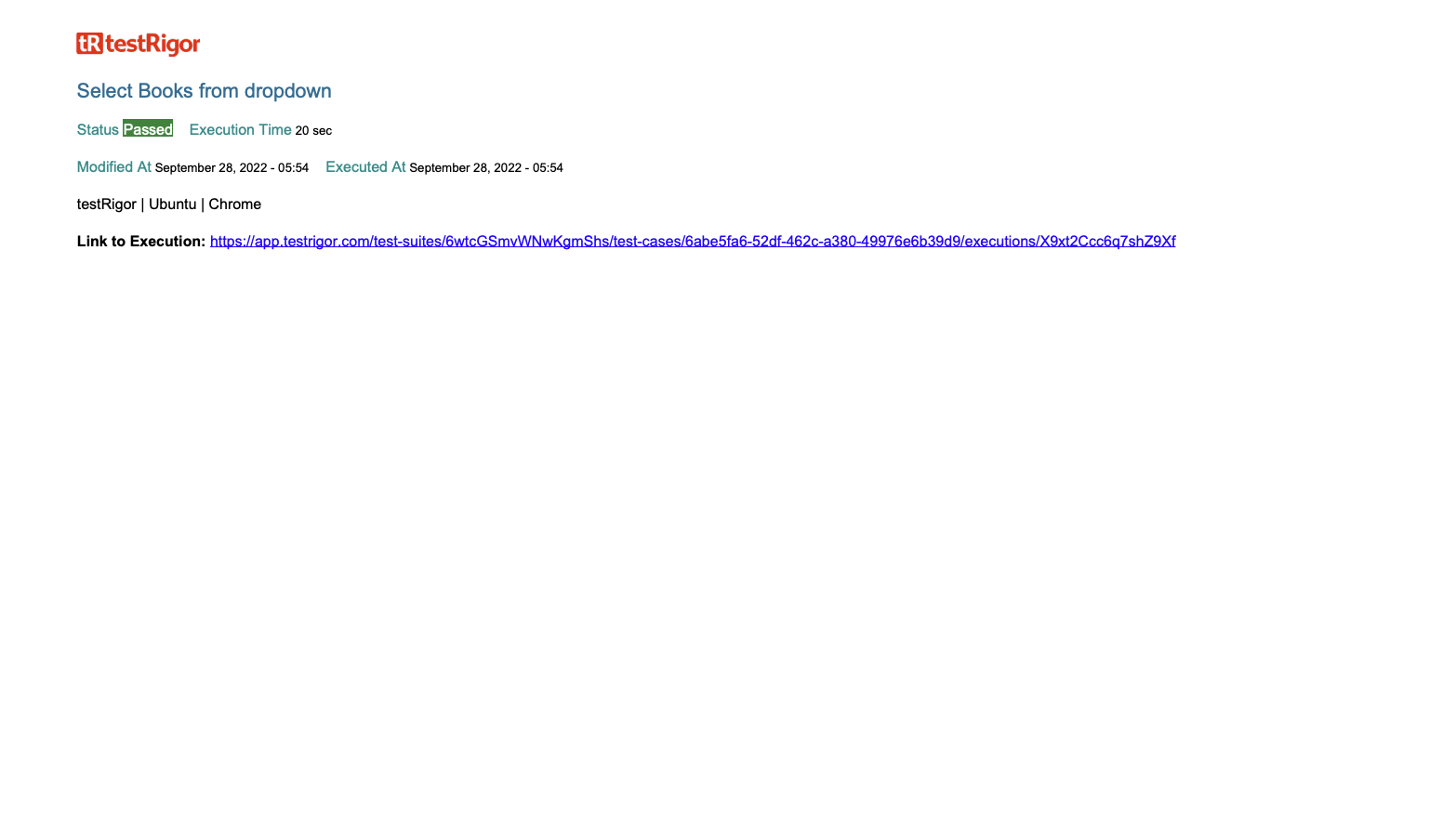
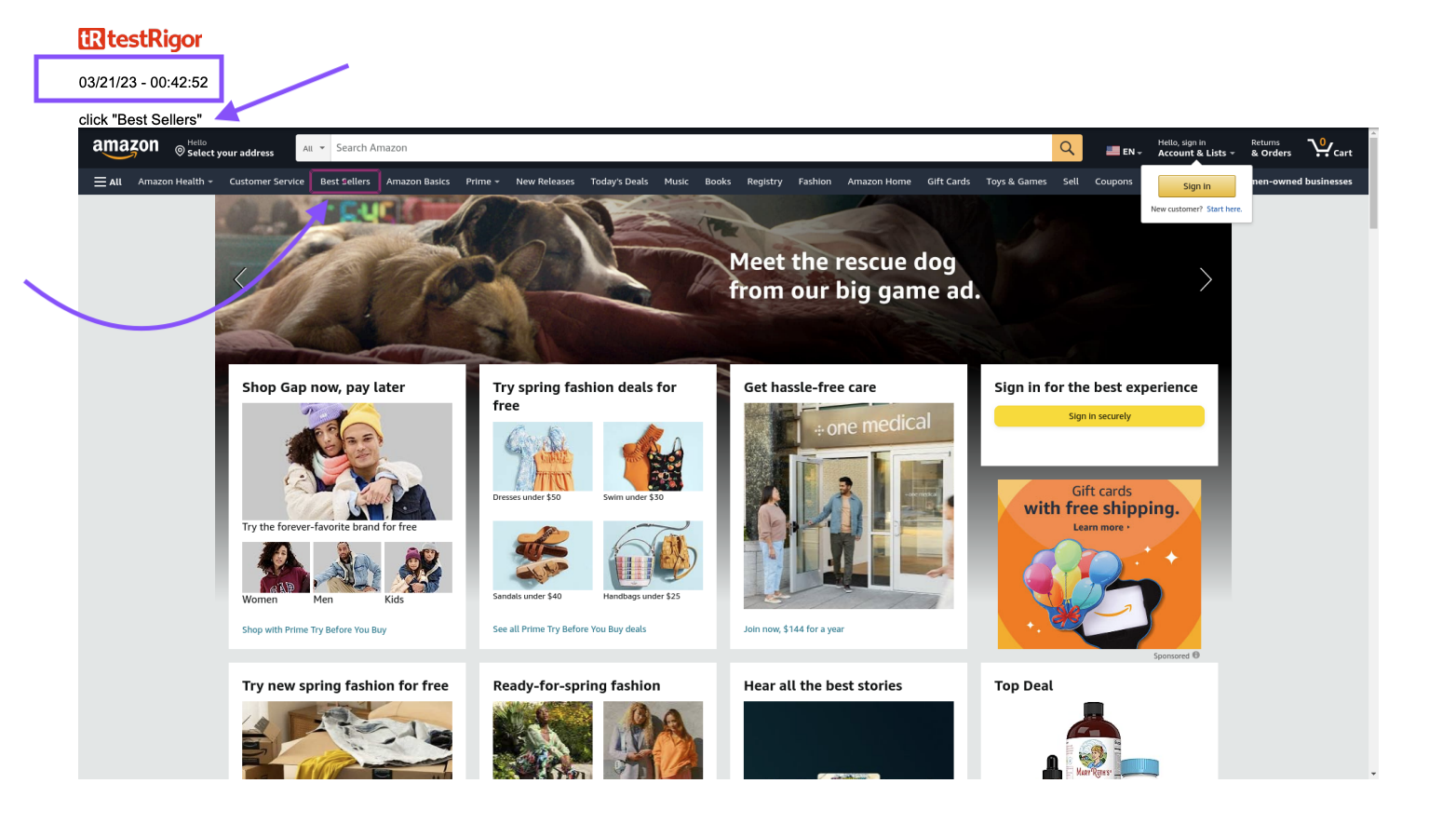
Conclusion
Incorporating 21 CFR Part 11 compliance is crucial for organizations operating within FDA-regulated industries that utilize electronic records and signatures. testRigor’s no-code test automation tool provides an efficient, user-friendly solution for organizations seeking to achieve 21 CFR Part 11 compliance. By automating key aspects of the compliance process, testRigor enables organizations to save time, reduce costs, and ensure the accuracy and reliability of electronic records and signatures. By integrating testRigor into their compliance strategy, organizations can successfully navigate the complex requirements of 21 CFR Part 11 and maintain long-term compliance with confidence.

